Case of the Month ...
Case History
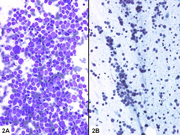
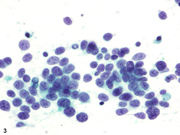
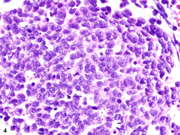
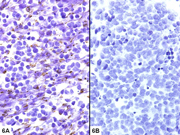
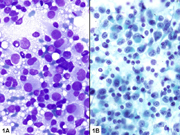
A 72 year old Caucasian male with multiple recurrences status post wide excision of malignant melanoma of the scalp presented with two new masses per PET scan, one left suboccipital (1.3 cm; Mass A ) and the other right axillary (1.5 cm; Mass B ). The patient has a chronic history of blistering sunburns, recently diagnosed prostate cancer, and no other known malignancy. He is a non-smoker and had a negative chest X-ray. The FNA of Mass A revealed metastatic melanoma (Fig. 1). The FNA results (smears and cell blocks; Figs. 2-6) of Mass B are shown below.
Diagnosis & Discussion
click on image for larger version
Image Figs:
- Figure 1- Mass A- (A) Diff-Quik stain- 400 x; (B) PAP stain- 400 x
- Figure 2- Mass B- (A) Diff-Quik stain- 200 x; (B) PAP stain- 200 x
- Figure 3- Mass B- PAP stain- 400 x
- Figure 4- Mass B- H&E stain- 400 x
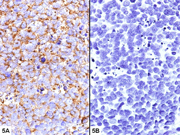
- Figure 5- Mass B- Immunostain (A) Synaptophysin- 400x; (B) HMB45- 400x
- Figure 6- Mass B- Immunostain (A) CK20- 400x; (B) TTF-1- 400x
Questions:
- What is the diagnosis?
- Metastatic malignant melanoma
- Chronic lymphocytic lymphoma
- Metastatic Merkel cell carcinoma
- Metastatic small cell lung cancer
- Which of the following statement correctly describes the cytological features of Mass B?
- Dispersed large, pleomorphic cells containing eccentric nuclei with single prominent nucleoli; widely spaced mirror-image binucleation can be seen
- Monomorphous small lymphocytes with clumped chromatin
- Predominantly single cells containing scant cytoplasm, and oval-to-round nuclei with fine granular chromatin
- Dispersed light and dark cells in a tigroid background; fine vacuolization can be seen in the cytoplasm
- Which immunophenotype is characteristic of Mass B?
- S-100, Mel-A, and HMB45 positive; PanKeratin and CD45 negative
- CD45, CD20, CD5, and CD23 positive; CD10 negative
- Synaptophysin, CK20, and neurospecific enolase positive; CK7 and TTF-1 negative
- Synaptophysin, CK7, TTF-1, and neurospecific enolase positive; CK20 negative
- Which of the following statement regarding Mass B is true?
- Recurrence and metastasis are both common
- Recurrence and metastasis are both extremely rare
- It is often indolent and carries a prognosis much better than that of cutaneous malignant melanoma
- Even though sometimes it initially presents as a metastasis to regional lymph node(s), a primary site is easy to be identified in all the cases.
Discussion:
Epidemiology and etiology
Merkel cell carcinoma (MCC) is a primary cutaneous neuroendocrine carcinoma first described in 1972 as trabecular carcinoma of the skin. Its origin from normal Merkel cells is controversial (1) . Although still rare (fewer than 2 cases per 100,000), MCC has its incidence tripled over the past 15 years. It primarily affects Caucasians, has a mean age at diagnosis of 69 years, and is slightly more common in males (60%). MCC most often occurs in sun exposed areas, with half of the cases in the head and neck.
Chronic sun exposure and immunosuppression have been considered risk factors for MCC. The incidence of MCC significantly increases after a diagnosis of certain other malignancies, including melanoma, cutaneous squamous cell carcinoma, basal cell carcinoma, chronic lymphocytic leukemia, and lymphomas (2) . Recently, a new polyoma virus, Merkel cell polyoma virus, has been found to clonally integrate at various sites in the human genome of the majority of MCC cases (3).
Clinical presentation, treatment, and prognosis
MCC often presents as a rapidly growing solitary asymptomatic pink-to-violet papule. About one third of the patients have regional lymph node involvement at diagnosis, and 10-20% patients present with nodal disease with unknown primary (4) . Metastasis is not uncommon, as skin, lymph nodes, liver, lung, and bone being the most frequent sites. Rare metastasis sites such as brain (5) , gastrointestinal tract (6) , and thyroid (7) have been also reported. The overall recurrence rate is over 50%. Spontaneous regression has been reported with an unknown mechanism.
Current management strategies include local excision, Mohs surgery, sentinel lymph node biopsy, lymph node dissection, irradiation, and chemotherapy. Despite of aggressive treatment, MCC carries a mortality rate around two times higher than that of melanoma, and about one third of the MCC patients will die from the disease (8).
Cytological findings and immunoprofiling
Histologically, MCC often presents as a poorly defined dermal mass infiltrating the subcutaneous soft tissue. Three histological patterns have been described, with no significant difference in prognosis: intermediate (the most common), small cell, and trabecular (1) . Cytologically, the aspirates are usually cellular, featuring predominantly dispersed small-to-intermediate sized cells with scant cytoplasm, oval-to-round nuclei with fine salt and pepper chromatin, numerous mitoses, and a high apoptotic index (1).
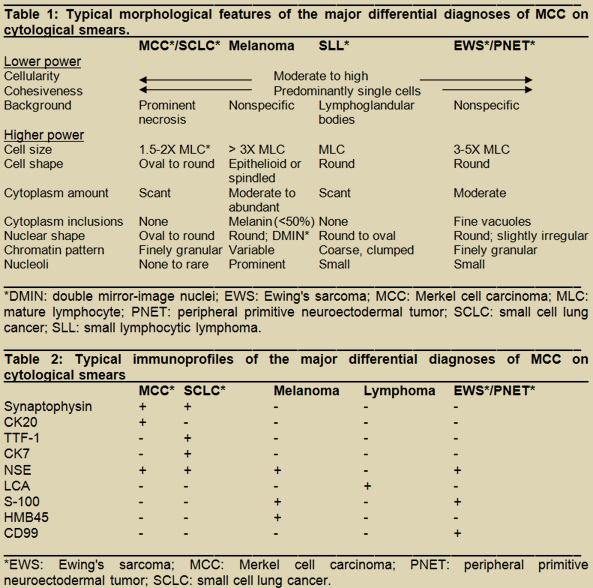
MCC falls in the small round blue cell category of tumors, in which the differential diagnosis includes other neuroendocrine tumors, melanoma, lymphoma, and EWS/PNET among others. In this reported case, melanoma needs to be carefully ruled out, especially because of the patient's history of multiple recurrences / metastases of melanoma, and the nature of melanoma as the great masquerader. Table 1 summarizes the typical morphological features of the major differentials of MCC on cytological smears. When high-power cytomorphology, patient age, genetic background, and clinical presentation often help rule out some differential diagnoses, MCC can be difficult to differentiate from other neuroendocrine tumors, especially small cell lung cancer (SCLC).
Immunoprofiling is helpful in the diagnosis of MCC, especially with an unknown primary, as in this reported case. CK20 is positive in over 80% of MCC cases, with a characteristic punctate perinuclear staining. CK20 positivity reduces when the cytoplasm is very scant or stripped. Table 2 summarizes the most commonly used markers in the major differentials of MCC on cytological smears. Exceptions do occur and worth noting. Rare cases of MCC that are TTF-1(+) (9) or CK7(+) (10) have been reported. Similarly, other entities in the differential list can also present with atypical immunophenotypes, which may overlap with that of MCC. A selected panel, rather a sole marker, is warranted, and a single deviated immunophenotype is insufficient to rule out MCC when its diagnosis is indicated by clinical and cytological features.
Answers
- c
- c
- c
- a
REFERENCES
Bichakjian CK, Lowe L, Lao CD, et al. Merkel cell carcinoma: critical review with guidelines for multidisciplinary management. Cancer 2007. 110:1-12.
Kaae J, Hansen AV, Biggar RJ, et al. Merkel Cell Carcinoma: Incidence, Mortality, and Risk of Other Cancers. J Natl Cancer Inst 2010 Apr 27 [Epub ahead of print].
Feng H, Shuda M, Chang Y, Moore PS. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science 2008. 319:1096-1100.
Jabbour J, Cumming R, Scolyer RA, Hruby G, Thompson JF, Lee S. Merkel cell carcinoma: assessing the effect of wide local excision, lymph node dissection, and radiotherapy on recurrence and survival in early-stage disease--results from a review of 82 consecutive cases diagnosed between 1992 and 2004. Ann Surg Oncol 2007. 14:1943-1952.
Feletti A, Marton E, Rossi S, et al. Pituitary metastasis of Merkel cell carcinoma. J Neurooncol 2010. 97:295-299.
Cheung M, Lee H, Purkayastha S, Goldin R, Ziprin P. Ileocaecal recurrence of Merkel cell carcinoma of the skin: a case report. J Med Case Reports 2010. 4:43.
Stoll L, Mudali S, Ali SZ. Merkel cell carcinoma metastatic to the thyroid gland: Aspiration findings and differential diagnosis. Diagn Cytopathol 2010 Jan 15 [Epub ahead of print].
Lemos B, Nghiem P. Merkel cell carcinoma: more deaths but still no pathway to blame. J Invest Dermatol 2007. 127:2100-2103.
Sierakowski A, Al-Janabi K, Dam H, Sood M. Metastatic Merkel cell carcinoma with positive expression of thyroid transcription factor-1--a case report. Am J Dermatopathol 2009. 31:384-386.
Pilloni L, Manieli C, Senes G, Ribuffo D, Faa G. Merkel cell carcinoma with an unusual immunohistochemical profile. Eur J Histochem 2009. 53:275-278.
Authors:
Xiangrong Zhao, MD
Department of Pathology and Laboratory Medicine
Berkshire Medical Center
725 North Street
Pittsfield, MA 01201*Nancy Caraway, MD
Department of Pathology, Section of Cytopathology
The University of Texas MD Anderson Cancer Center
1515 Holcombe Blvd
Houston, TX 77030*Corresponding to:
Nancy Caraway, MD
Department of Pathology, Section of Cytopathology
The University of Texas
MD Anderson Cancer Center, Box 53
1515 Holcombe Blvd
Houston, TX 77030
Tel: (713) 792-2068
Fax: (713) 792-2313